Medical Terminology Daily (MTD) is a blog sponsored by Clinical Anatomy Associates, Inc. as a service to the medical community. We post anatomical, medical or surgical terms, their meaning and usage, as well as biographical notes on anatomists, surgeons, and researchers through the ages. Be warned that some of the images used depict human anatomical specimens.
You are welcome to submit questions and suggestions using our "Contact Us" form. The information on this blog follows the terms on our "Privacy and Security Statement" and cannot be construed as medical guidance or instructions for treatment.
We have 1819 guests online

Marcia Crocker Noyes
(1869 – 1946)
Further to my comment on old books and research that started with an interesting bookplate (Ex-Libris). I continued my research and found that the person in charge of the Osler library bookplate was a fascinating individual that today maybe a ghost in the MedChi library and building in Baltimore... This is certainly an article that can be called "A Moment in History"
Marcia Crocker Noyes was the librarian at The Maryland State Medical Society from 1896 to 1946 and was a founding member of the Medical Library Association.[1][2][3]
Sir William Osler, MD. a famous Johns Hopkins surgeon was a noted bibliophile and had a large personal collection of books on various topics. When he became the President of MedChi in 1896, he was dismayed at the condition of the library and knew that with the right person and some stewardship, it could become a significant collection. Sir William asked his friend, Dr. Bernard Steiner, a physician and President of the Enoch Pratt Free Library in Baltimore for suggestions of a librarian, and Dr. Steiner recommended Marcia Crocker Noyes. A native of New York, and a graduate of Hunter College, Marcia had moved to Baltimore for a lengthy visit with her sister, and took a “temporary” position at the Pratt Library, which turned into three years. Although she had no medical experience or background, she was enthusiastic, and most importantly, she was willing to move into the apartment provided for the librarian, who needed to be available 24 hours a day.
The image in this article is Ms. Noyes on her first year on the job. Marcia developed a book classification system for medical books, based on the Index Medicus, and called it the Classification for Medical Literature. The system uses the alphabet with capital letters for the major divisions of medicine and lower-case ones for the sub-sections. The system was used for many years, but it's now dated and the Faculty's original shelving scheme was never changed. The card catalogs still reflect her classification and many of the cards are written in Marcia's back-slanting handwriting.
Marcia knew enough to ask the Faculty's members about medical questions, terminology and literature. She gradually won over the predominantly male membership and they became her greatest allies; Sir William at the start, and then for nearly 40 years, Dr. John Ruhräh, a wealthy pediatrician with no immediate family of his own. She made a point of attending almost every Faculty function, and in 1904, under guidelines from the American Medical Association, Marcia was made the Faculty Secretary. For much of her first 10 years, she was the Faculty's only full-time employee, only being assisted by Mr. Caution, the Faculty's janitor. Later in life Marcia would say that she hired him because of his name!
Within ten years, the library had outgrown its space, and plans, spearheaded by Marcia and Sir William before his move to Oxford, were made to build a headquarters building, mainly to house the library's growing collection of medical books and journals.
Marcia was instrumental in the design and building of the new headquarters. She travelled to Philadelphia, New York and Boston to look at their medical society buildings, and eventually, the Philadelphia architectural firm, Ellicott & Emmart was selected to design and build the new Faculty building. Every detail of the building held her imprimatur, from the graceful staircase, to the light-filled reading room, and all of the myriad details of the millwork, marble tesserae, and most of all, the four-story cast iron stacks. She was on-site, climbing up unfinished staircases, checking out the progress of the building, which was built in less than one year at a cost of $90,000.
Among the features of the new building was a fourth-floor apartment for her. She referred to it as the "first penthouse in Baltimore" and it had a garden and rooftop terrace. The library collection eventually grew to more than 65,000 volumes from medical and specialty societies around the world. Journals were traded back and forth, and physicians eagerly anticipated the arrival of each new issue. At the same time, Marcia was involved in the Medical Library Association as one of eight founding members. The MLA promotes medical libraries and the exchange of information. One of the earliest mandates of the MLA was the Exchange, a distribution and trade service for those who had duplicates or little-used books in their collections. Initially, the Exchange was run out of the Philadelphia medical society, but in 1900 it was moved to Baltimore and Marcia oversaw it. Several hundred periodicals and journals were received and sent each month, a huge amount of work for a tiny staff. In 1904, the Faculty had run out of room to manage the Exchange, so it was moved to the Medical Society of the Kings County (Brooklyn). But without Marcia's excellent administrative skills, it floundered and in 1908, the MLA asked Marcia to take charge once again.
In 1909, when the new Faculty building opened, there was enough room to run the Exchange and with the help of MLA Treasurer, noted bibliophile and close friend, Dr. John Ruhräh, it once again became successful. Additionally, Marcia and Dr. Ruhräh combined forces to revive the MLA's bulletin, which had all but ceased publication in 1908, taking the Exchange with it. This duo maintained editorial control from 1911 until 1926. In 1934, around the time of Dr. Ruhräh's death, Marcia became the first “unmedicated” professional to head the MLA. During her tenure, the MLA incorporated, the first seal was adopted, and the annual meeting was held in Baltimore. Marcia wanted to write the history of the MLA once she retired from full-time work at the Faculty, but her health was beginning to fail. She had back problems and had suffered a serious burn on her shoulder as a young woman, possibly from her time running a summer camp, Camp Seyon, for young ladies in the Adirondack Mountains. In 1946, a celebration was planned to honor Marcia's 50 years at the Faculty. But she was adamant that the physicians wait until November, the actual date of her 50 years. However, they knew she was gravely ill, and might not make it until then, so a huge party was held in April. More than 250 physicians attended the celebration, but the ones she was closest to in the early years, were long gone. She was presented with a suitcase, a sum of money to use for travelling, and her favorite painting of Dr. John Philip Smith, a founder of the Medical College in Winchester, Virginia. It was painted by Edward Caledon Smith, a Virginia painter who had been a student of the painter Thomas Sully.[4] She adored this painting and vowed, jokingly, to take it with her wherever she went.
The painting was not to stay with her for very long, for she died in November 1946, and left it to the Faculty in her will. Her funeral was held in the Faculty's Osler Hall, named for her dear friend. More than 60 physicians served as her pallbearers, and she was buried at Baltimore's Green Mount Cemetery. In 1948, the MLA decided to establish an award in the name of Marcia Crocker Noyes. It was for outstanding achievement in medical library field and was to be awarded every two years, or when a truly worthy candidate was submitted. In 2014, the Faculty began giving a bouquet of flowers to the winner of the award in Marcia's name, and in honor of her work. Much evidence exists for this tradition, as we know that the physicians, especially Drs. Osler and Ruhräh, frequently gave her bouquets of flowers. Marcia also cultivated flower gardens at the Faculty and decorated the rooms with her work.
Today, the MedChi building is open for tours and if the rumors are to be believed Ms. Marcia Crocker Noyes is still at work in her beloved library as the "resident ghost" [1][5]
NOTE: This article has been modified from the original Wikipedia article on Marcia Crocker Noyes. The article itself is well-written with interesting images of the subject. I would encourage you to visit it. The second insert is from book 00736 in my personal library and shows in pencil, the incredibly small handwriting of Marsha C. Noyes.
Sources:
1. "Marcia, Marcia, Marcia" MedChi Archives blog.
2. "Marcia C. Noyes, Medical Librarian" (PDF). Bulletin of the Medical Library Association. 35 (1): 108–109. 1947. PMC 194645
3. Smith, Bernie Todd (1974). "Marcia Crocker Noyes, Medical Librarian: The Shaping of a Career" (PDF). Bulletin of the Medical Library Association. 62 (3): 314–324. PMC 198800Freely accessible. PMID 4619344.
4. Edward Caledon BRUCE (1825-1901)"
5. Behind the scenes tour MedChiBuilding
"Clinical Anatomy Associates, Inc., and the contributors of "Medical Terminology Daily" wish to thank all individuals who donate their bodies and tissues for the advancement of education and research”.
Click here for more information
- Details
- Written by: Efrain A. Miranda, Ph.D.
This article is part of the series "A Moment in History" where we honor those who have contributed to the growth of medical knowledge in the areas of anatomy, medicine, surgery, and medical research.
In late March 2026, I was reading an article on the Journal of Clinical Anatomy written by R. Shane Tubbs PhD. MS, PA-C, past president of the American Association of Clinical Anatomists. The article was titled “Doctors Without Anatomy Are Like Moles: They Work in the Dark…”.
He was referring to a dictum by Friedrich Tiedeman, MD. His original dictum was:
“Ärzte ohne Anatomie gleichen Maulwürfen: sie arbeiten im Dunkeln, und ihrer Hände Tagewerk sind — Erdhügel”
The translation is in the first image in this post. Dr. Tubbs journal article led me to research Tiedemann’s life, scientific work, and legacy, where besides his famous sentence (which is very common use in Germany medical schools) I found a correlation between Tiedemann' work and a famous energy drink!
Friedrich Tiedemann (1781–1861) was a German physician, anatomist, physiologist, and zoologist. Born in Kassel, Germany. He studied medicine at the University of Marburg, where he graduated with an MD in 1804. Soon after graduation he abandoned medical practice and dedicated himself to scientific research and academic teaching, focusing particularly on anatomy and physiology.
At the University of Marburg, he taught physiology and comparative osteology. In 1805, at only twenty-four years of age, he was appointed Professor of Zoology, Human Anatomy, and Comparative Anatomy at the University of Landshut.
Later, in 1816 he took the chair of anatomy and physiology at the University of Heidelberg until his retirement in 1854. During these three decades he served as director of the anatomical institute, including various university administrative roles including dean and prorector. He took retirement because of increasing blindness, and he was forced to sell his library which contained thousands of books,
During his tenure he expanded the anatomical collections substantially. By the time of his retirement in 1849, the institute contained more than 2,000 anatomical and pathological specimens, including injected vascular preparations and comparative zoological material used for teaching and research.
Tiedemann was elected to several scientific societies, including:
• Royal Society of London
• Royal Swedish Academy of Sciences
• American Academy of Arts and Sciences
• Royal Society of Edinburgh
Tiedemann’s early research focused on the development and comparative structure of the brain. His work "Anatomie und Bildungsgeschichte des Gehirns im Foetus des Menschen" (Anatomy and developmental history of the brain in the human fetus ), published in 1816., examined embryonic brain development and compared human brain structures with those of animals, providing insights into morphological development.
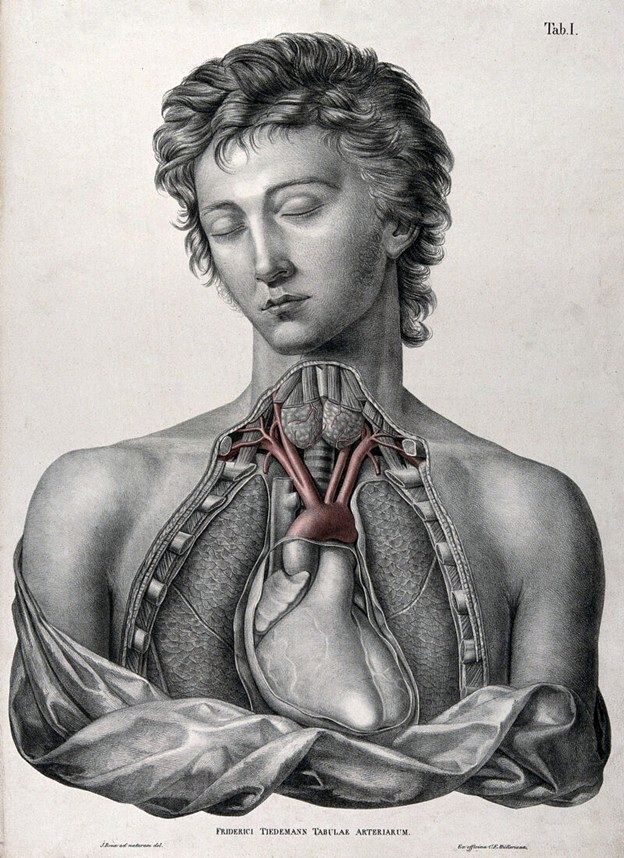
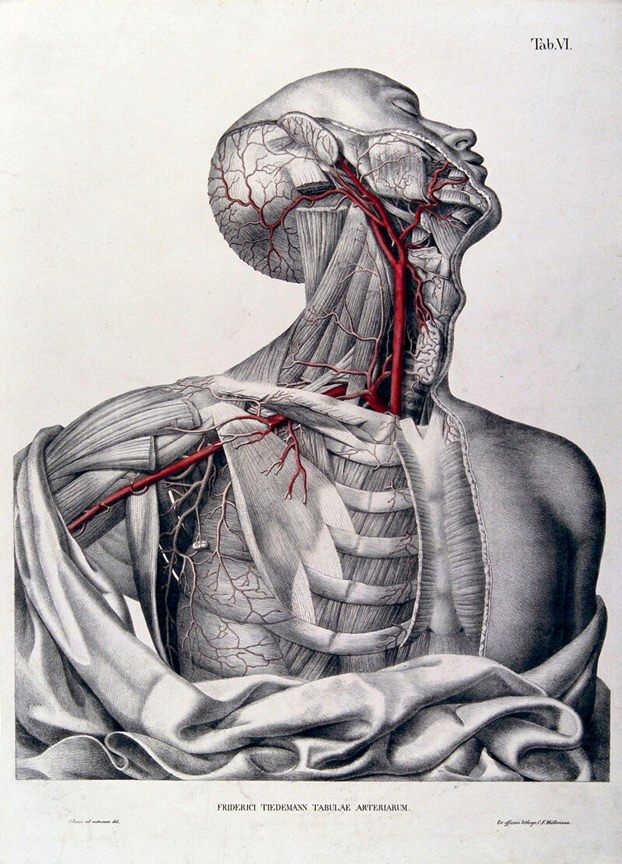
He authored several publications, but probably the most famous was his work “Tabulae Arteriarum Corporis Humani” (Plates of the arteries of the human body) published in 1822 with illustrations by Jacob Wilhelm Christian Roux is a folio-size anatomical atlas devoted entirely to the arterial system. The atlas has 38 large lithographic plates, many originally hand-colored, making it one of the earliest works devoted exclusively to the arteries of the human body. This book is in two different volumes, one with the images (available here) and one with the listing of the structures in both Latin and German (available here)
He was one of the first to challenge racial anthropology, arguing with scientific facts, that social inequalities were products of historical and political circumstances, including slavery, rather than biological differences. Among Tiedemann’s most historically notable publications was “On the Brain of the Negro, Compared with That of the European and the Orang-Outang” (1836–1837)", published in the Philosophical Transactions of the Royal Society.
An interesting side of Tiedemann’s research was the result of his research on the physiology of digestion. In collaboration with the chemist Leopold Gmelin (1788–1853), Tiedemann conducted experimental studies on digestion and metabolism. Their work “Die Verdauung nach Versuchen" (Experiments on Digestion- published on 1827) examined the physiological mechanisms of digestion and helped confirm the role of chemical processes in gastric activity. They analyzed the composition of bile in the bull (Lat: Taurus) and discovered one of its main components (2-aminoethanesulfonic acid) which they called “Taurine” a word derived from the Latin "taurus" meaning bull, because it was discovered in the bull gallbladder and intestine.
Although not an essential dietary component, this compound is used today as a dietary supplement and, believe it or not, it is one of the components of an energy drink … Red Bull! Talk about coincidences!
Sources:
1. Tubbs, R.S. (2026), Doctors Without Anatomy Are Like Moles: They Work in the Dark…. Clinical Anatomy
2. LaTourelle J. Friedrich Tiedemann (1781–1861). Embryo Project Encyclopedia. Arizona State University; 2015.
3. University of Heidelberg Library. Friedrich Tiedemann (1781–1861) digital collections overview. (ub.uni-heidelberg.de)
4. Encyclopaedia Britannica (9th ed.). Tiedemann, Friedrich.
5. Heirs of Hippocrates Collection, University of Iowa Libraries. Tabulae arteriarum corporis humani bibliographic record.
6. Encyclopedia.com. Tiedemann, Friedrich (1781–1861).
7. Royal Society. Friedrich Tiedemann—Fellowship record.
Image of Dr. Tiedemann public domain, AI enhanced for color. Images of Tiedemann's book public domain.
- Details
- Written by: Efrain A. Miranda, Ph.D.
- Hits: 5680
This article is part of the series "A Moment in History" where we honor those who have contributed to the growth of medical knowledge in the areas of anatomy, medicine, surgery, and medical research.
Karel Frederik Wenckebach (1864–1940) Dutch physician and anatomist born in 1864, in The Hague, Netherlands. Wenckebach enrolled at the University of Utrecht’s School of Medicine, obtaining his degree in 1888.
While at medical school, he developed interest in physiological research, training in laboratory techniques and cardiac rhythm analysis under the mentorship of physiologist Theodor Wilhelm Engelmann (1843 -1909). His early work focused on embryology and rhythm disturbances observed in isolated frog hearts—a critical foundation for his later discoveries in human cardiac conduction and cardiac arrhythmias.
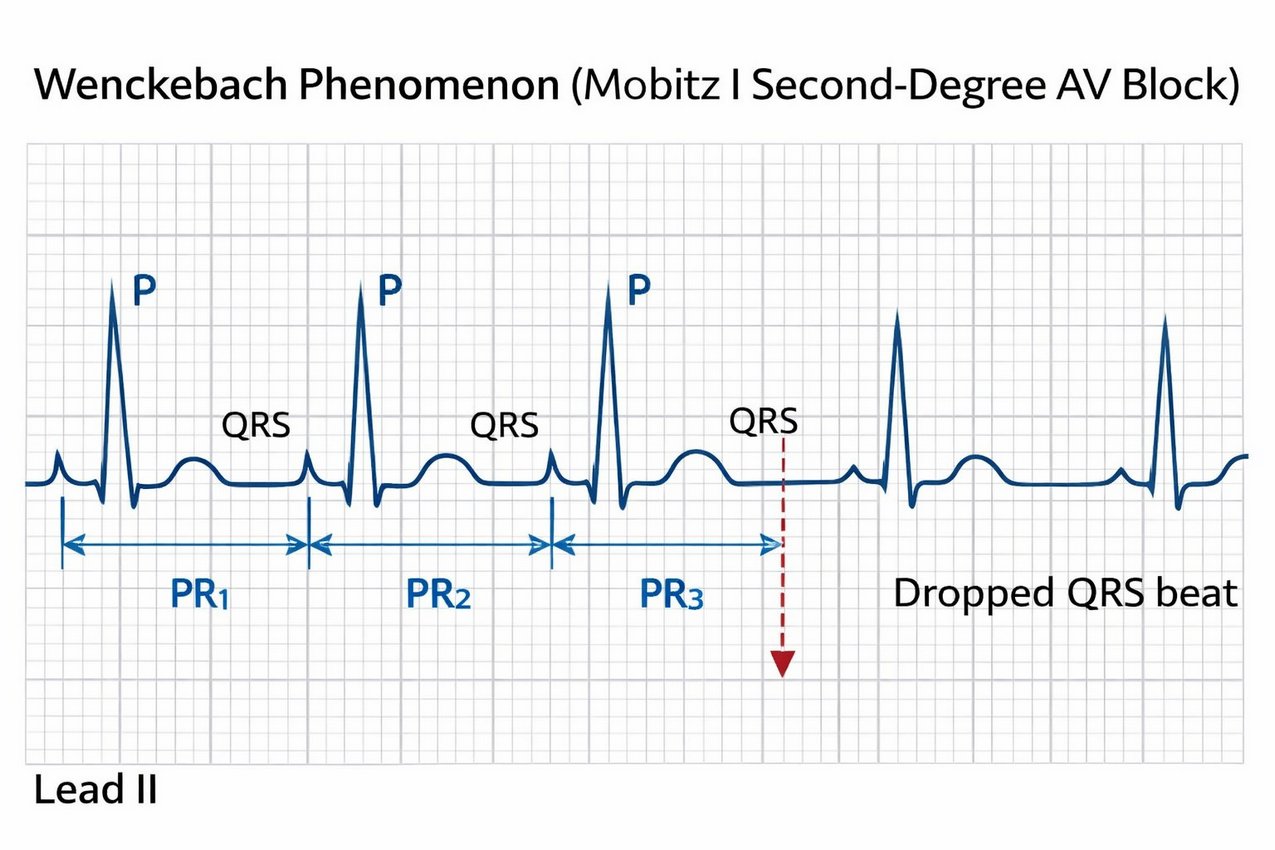
After graduation, Wenckebach briefly practiced medicine in rural Netherlands, where he encountered patients with irregular heartbeats. Through careful observational analysis of arterial pulse tracings—without the benefit of electrocardiographs—he identified a pattern of progressive prolongation in pulse intervals followed by a “dropped beat.” In 1899, he described this arrhythmic sequence, initially termed "Luciani periods", that later came to be recognized as second-degree atrioventricular (AV) block, Mobitz type I, or the Wenckebach phenomenon.
The pattern of this arrhythmia reflected a progressive delay in conduction through the AV node until one impulse failed to be conducted, resetting the cycle. Wenckebach’s work provided early evidence that conduction properties intrinsic to the myocardium and its specialized tissues underlay certain rhythm disturbances rather than ectopic beats alone.
Wenckebach’s insight was notable for preceding the advent of clinical electrocardiography and the anatomical identification of the sinoatrial (SA) node and the atrioventricular (AV) node by Tawara, Keith and Flack and other researchers in the early 20th century.
In addition to describing rhythm phenomena, Wenckebach contributed to early understanding of internodal conduction pathways. In his later work, he described an anatomical tract—now referred to as Wenckebach’s bundle or the middle internodal tract—as part of the internodal conduction system that links the SA node to the AV node.
Wenckebach’s academic career included professorships at the University of Groningen (1901), University of Strasbourg (1911–1914), and University of Vienna (1914–1929). He authored influential texts including Die Arhythmie als Ausdruck bestimmter Funktionsstörungen des Herzens (1903) and Die unregelmässige Herztätigkeit und ihre klinische Bedeutung (1914), which integrated clinical observations with physiological concepts. Wenckebach also investigated pharmacological interventions in arrhythmia, notably promoting the use of quinine for paroxysmal atrial fibrillation.
His contributions earned numerous honors, including honorary fellowships and international recognition. Wenckebach retired in 1929 and continued to study cardiac physiology, including investigations on heart failure in the Dutch East Indies. He died in Vienna on November 11, 1940.
Dr. A.R. Perez-Riera (Brazil) quotes Wenckeback as follows:
“In medical science there are vast realms of which I have no special knowledge and, again, no, I am not a great man; I am a happy man.”
Sources and references:
1. Wenckebach KF. Die Arhythmie als Ausdruck bestimmter Funktionsstörungen des Herzens. Leipzig: Verlag; 1903.
2. Upshaw CB Jr. The Wenckebach phenomenon: a salute and comment on the centennial of its original description. Ann Intern Med. 1999;131(8):634. PubMed PMID: 9890852.
3. Mendoza-Davila N, Varon J. Resuscitation great. Karel Wenckebach: the story behind the block. Resuscitation. 2008;79(2):189-192.
4. Cadogan M. Karel Frederick Wenckebach. LITFL Medical Eponym Library. 2025.
5. Pérez-Riera AR, et al. Median bundle of Wenckebach and internodal conduction pathways in cardiac anatomy. Via Medica (PDF).
6. Zhao Y, Wan J, Liao B, Qi M. The Neglected Internodal Tract-A Cardiac Conduction System Structure Homologous to the Development and Regulation of the Sinoatrial Node. Rev Cardiovasc Med. 2025 Apr 22;26(4):27882. doi: 10.31083/RCM27882. PMID: 40351691; PMCID: PMC12059794.
7. José L. Fresquet. Karel Frederik Wenckebach (1868-1940) Instituto de Historia de la Ciencia y Documentación (Universidad de Valencia-CSIC). Marzo, 2010.
Image of Dr. Wenckebach public domain, AI enhanced for color.
- Details
- Written by: Efrain A. Miranda, Ph.D.
- Hits: 1982
Walter Eduard Carl Koch (1880–1962), German physician, anatomist, and pathologist. Born in Dortmund, Germany, studied medicine at the University of Freiburg and the Kaiser Wilhelm Academy in Berlin, attaining his doctorate in 1907.
He served as a military physician, and later at the Kaiser Wilhelm Academy and in Berlin. Koch specialized in pathology and pathological anatomy and was appointed professor in 1922. Later he became head of the Pathology Department at the Westend Hospital in Berlin and held academic positions after World War II, including a professorship at the newly reconstituted Free University of Berlin. He died in 1962.
He is remembered by the eponymous “triangle of Koch, “site for the atrioventricular node. Although he never referred to this area as a triangle, or used his name to refer to this area, he described it as we know it today in his 1922 publication “Der Funktionelle Bau des Menschlichen Herzens” (The functional structure of the human heart).
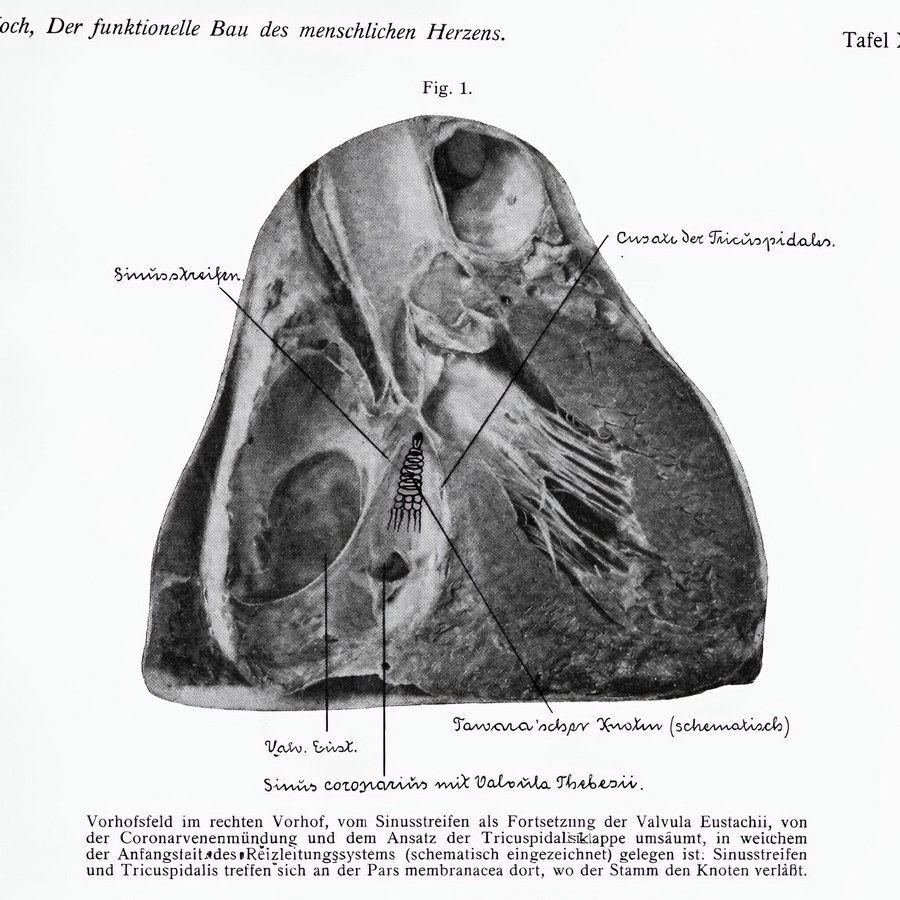
Plate X.1 by Walter EC Koch (1922)
In Plate 10.1 Koch shows a photograph of the right atrium where he has drawn the “Tawara’schen Knotten”- an early name for the atrioventricular node (knot of Tawara) – and he describes it as follows: “Vorhofsfeld im rechten Vorhof, vom Sinusstreifen als Fortsetzung der Valvula Eustachii, von der Coronarvenenmündung und dem Ansatz der Tricuspidalisiklappe umsäumt, in weichem der Anfangstait des Reizleitungssystems (schematisch eingezeichnet) gelegen ist: Sinusstreifen und Tricuspidalis treffen sich an der Pars membranacea dort, wo der Stamm den Knoten verläßt” (The atrial region in the right atrium, bordered by the sinoatrial band, a continuation of the Eustachian valve, by the coronary vein (sinus) orifice and the insertion of the tricuspid valve, in which the initial stage of the conduction system (schematically shown) is located: the sinoatrial sinus and the tricuspid valve meet at the pars membranacea where the trunk leaves the node.) The handwriting on the image is by Koch himself.
Note that Koch does not mention the tendon of Todaro as a landmark in this description (although Todaro’s tendon is mentioned in this publication), and Koch calls it the “sinoatrial band").
The term "triangle of Koch" refers to a triangular area in the right atrium with defined anatomical boundaries:
• Medial border: the tendon of Todaro
• Inferior border: the tricuspid valve leaflet
• Posterior border: the orifice of the coronary sinus.
The apex of this triangle corresponds to the location of the atrioventricular node.
Note: In spite of my efforts, I could not find a photograph or a portrait of Walter E.C. Koch. Dr. Miranda.
Sources:
1. "Remembering the canonical discoverers of the core components of the mammalian cardiac conduction system: Keith and Flack, Aschoff and Tawara, His, and Purkinje" Icilio Cavero and Henry Holzgrefe Advances in Physiology Education 2022 46:4, 549-579.
2. "Tratado de Anatomia Humana" Testut et Latarjet 8th Ed. 1931 Salvat Editores, Spain
3. Anderson RH, Sánchez-Quintana D, Nevado-Medina J, Spicer DE, Tretter JT, Lamers WH, Hu Z, Cook AC, Sternick EB, Katritsis DG. . The Anatomy of the Atrioventricular Node. J of Cardiovasc Development and Disease. 2025; 12(7):245.
4. Koch, Walter “Der Funktionelle Bau des Menschlichen Herzens” Urban & Schwarzenberg. 1922. Berlin
- Details
- Written by: Efrain A. Miranda, Ph.D.
- Hits: 1954
This article is part of the series "A Moment in History" where we honor those who have contributed to the growth of medical knowledge in the areas of anatomy, medicine, surgery, and medical research.

Francesco Todaro
Francesco Todaro (1839 – 1918) Italian physician and anatomist born in Tripi, a village in the province of Messina, Sicily.
While still a medical student at the University of Messina, he joined the Garibaldini as a volunteer and fought at Corriolo and Milazzo during the campaign for Italian unification in 1860. After completing his medical degree, he pursued further anatomical training in Florence, studying under prominent figures including Moritz Schiff and Filippo Pacini (1812 – 1883) focusing his research myocardial and valve anatomy.
In his 1865 publication, "Novelle ricerche sopra la struttura delle orecchiette del cuore umano e sopra la valvola di Eustachio" Todaro described a fibrous extension of the valve of the inferior vena cava (Eustachian valve) starting at the point where the Eustachian valve and the valve of the coronary sinus (Thebesian valve) appear to merge. This structure later became known eponymically as the tendon of Todaro, component of the boundaries of the triangle of Koch, site for the atrioventricular node.
In page 33 of this publication Todaro states: “e perseguitatolo, con ogni accuratezza, in mezzo alle fibre muscolari del sotto, l'accompagnai fino al pilastro posteriore della fossa ovale, da ove mi accertai entrare pel corno posteriore, che qui ha origine, nella valvula di Eustachio, percorrendola in tutto il suo margine libero. Ho confermato dopo, questa osservazione, in molti altri casi ; ed ho trovato sempre costante, il tendine, anco quando la valvula d'Eustachio è quasi manchevole.” “I followed it to the posterior pillar of the oval fossa, from where I confirmed that it entered through the posterior horn, which originates here, into the Eustachian valve, following its entire free margin. I have subsequently confirmed this observation in many other cases, and have always found the tendon to be constant, even when the Eustachian valve is almost missing.”
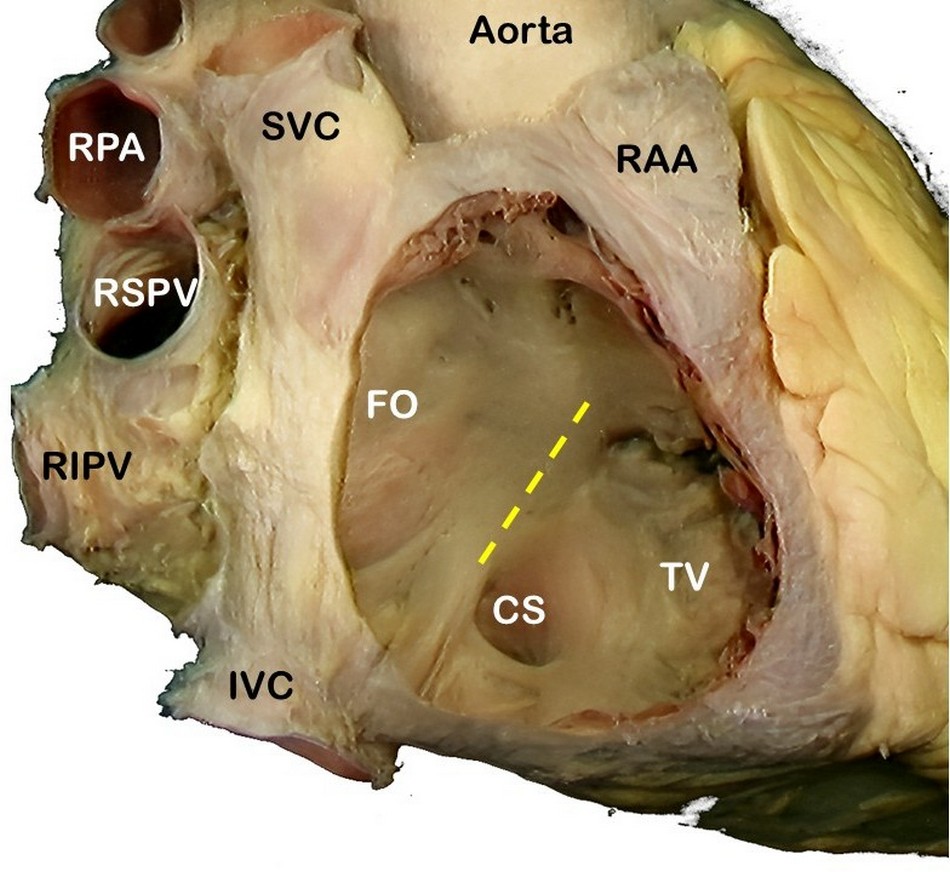
SVC: Superior vena cava, RPA: Right pulmonary artery, RSPV: Right superior pulmonary vein, RIPV: Right inferior pulmonary vein, IVC: Inferior vena cava, RAA: Right atrial appendage, FO: Foramen ovale, TV: Septal leaflet of the tricuspid valve. The yellow line shows the location of the "Tendon of Todaro".
In 1867 he was appointed Professor of Human Anatomy at the University of Messina, and in 1871 he accepted the chair of anatomy at the University of Rome “La Sapienza”, where he remained until his death.
In 1874, Todaro became a member of the Accademia Nazionale dei Lincei, Italy’s foremost scientific academy. He also served as a Senator of the Kingdom of Italy.
Personal note: There are some publications that assert that the tendon of Todaro is rarely seen. That has not been my experience.
Sources:
1. Sopra la struttura delle orecchiette del cuore umano. Todaro F. Stabilimento Civelli Pub. Florence, Italy: 1865. (Italian)
2. TODARO, Francesco. Dorello P. Enciclopedia Italiana. 1937 (Italian)
3. Todaro, Francesco. Treccani Enciclopedia on line.(Italian)
4. TODARO, Francesco. Ottaviani A. Dizionario Biografico degli Italiani. Vol. 95. (Italian)
5. Cardiac anatomy primer. McMullen, HL. Am Assoc Thorac Surg.
- Details
- Hits: 6014
This article is coauthored by Randall K. Wolf, MD and Efrain A. Miranda, PhD
Can there exist a unified theory that deciphers the mechanisms for cardiac arrythmias and atrial fibrillation (AFib) that can also explain the results of various catheter-based and surgical-based treatments? Based on anatomical and physiological study of the heart and it’s nervous system, we believe the answer is yes.
While we have learned not to expect everyone to be persuaded by the argument we will be presenting, we suggest, nevertheless, this argument does deserve careful and dispassionate consideration for it provides a model which explains results in the treatment of AFib, and should not be ignored. The epiphany that shed light on a possible unifying mechanism came during multiple cases of left atrial electrical testing during minimally invasive surgical pulmonary vein isolation (PVI) in patients who had multiple previous catheter-based PVI ablation procedures.
Over the years we have evolved our view of the anatomical structures and processes that control the beating rhythmic activity of the heart. Additional and complementary information can be found following the links in the article.
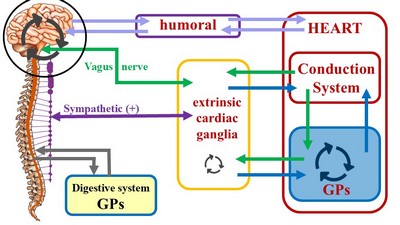
First, we need to define two terms in reference to the heart: “Intrinsic” and “Extrinsic”. An intrinsic structure is found within the boundaries of the heart, that is, within the parietal pericardium. Extrinsic means that the structure about is found outside the parietal pericardium.
The most well-known intrinsic component is what we know as “the conduction system of the heart”. The classic description of the conduction system of the heart emphasizes this cardiomyocyte- based component and refers to a group of specialized cardiac muscle structures that serve as pacemakers and distributors of the electrical stimuli that make the heart beat coordinately. It is important to stress the fact that this primary "conduction system of the heart" is not formed by nerves, but rather by specialized cardiac muscle cells. Probably the reason so much emphasis is placed on the conduction system of the heart is the use of the electrocardiogram as a clinical diagnostic tool since William Einthoven (1860 - 1927) introduced the EKG in 1901.
The second (quite complex) component of this system has been forced to take a secondary place, and in many cases ignored. We refer to the modulating activity on the heart by the autonomic nervous system (ANS), with its two subsystems, sympathetic and parasympathetic.
Unlike the conduction system of heart, which is purely intrinsic, the cardiac autonomic system is both extrinsic and intrinsic. The classic architecture described is a two-neuron system, where an extrinsic preganglionic neuron located in the central nervous system (CNS) connects with an extrinsic postganglionic neuron found in the sympathetic chain and ganglia located in the superficial and deep cardiac plexuses close to the aorta and pulmonary trunk.
The cardiac ANS has an intrinsic component formed by ganglionated plexi (GPs) and nerves found within the walls of the myocardium and in epicardial areas of the heart which contain fat. The main location of these GPs is close to and around the great vessels. These ganglionated plexi are the basis for the complex rhythmicity responses of the hear. In fact, several researchers call these intracardiac plexuses the "Little Brain of the Heart". Failure of the ganglionated plexi are the basis of many cardiac arrythmias, including atrial fibrillation.
The inclusion of the cardiac ganglionated plexi into this picture has led us to propose a different ANS organization for those organs that have rhythmicity, be it a beat (heart) or peristalsis (digestive system, ureters, urethra, etc.). For a more detailed explanation follow this link to another article on this topic.
These cardiac intrinsic ANS component are responsible for the complex reflexes that increase or decrease both the heartbeat and the force of contraction of the heart muscle in response to variations in volumetric pressure in the atria and chemical variations in the blood caused by alcohol, caffeine, drugs, dehydration, etc. A more detailed explanation can be found in the following Houston Methodist DeBakey CV Live video. Click on the video to start it or you can go directly to YouTube by clicking here. The main content start at 2 minutes:
The cardiac ANS has important communication with the brain centers responsible for mood and emotions. It is important to note that emotional response is linked to visceral activity (glands and viscera), and this includes the heart.
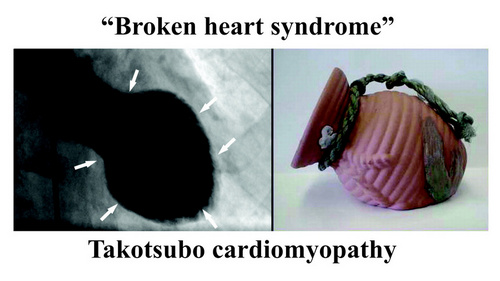
Also, the ANS/GPs complex is not separated from the higher functions of the forebrain. Cechetto (2005) explains how the forebrain (conscious) activity influences the spinal cord and the ANS by pathways that include the limbic system, insula, amygdala, and lateral hypothalamus. These pathways and communications can certainly explain arrythmias caused by stress and anxiety and pathologies such as the “broken heart syndrome” (Takotsubo cardiomyopathy).
Note: "Broken heart syndrome" or "stress cardiomyopathy" is also known as "Takotsubo cardiomyopathy". This is because the shape of the heart in this condition changes and resemble a Japanese octopus (tako) trap (tsubo). For more information on this condition, click here.
Sources and references
1. Kawashima, T. The autonomic nervous system of the human heart with special reference to its origin, course, and peripheral distribution. Anat Embryol (2005) 209: 425–438
2. D. F. Cechetto, "Forebrain Control of Healthy and Diseased Hearts," Chapter in "Basic and Clinical Neurocardiology", J. Armour and J. L. Ardell, Eds., Oxford University Press, 2004.
3. Kandel ER, Koester JD, Mack SH, Siegelbaum SA. Principles of Neural Science. 6th ed. New York, NY: McGraw Hill; 2021.
4. Standring S, ed. Gray’s Anatomy: The Anatomical Basis of Clinical Practice. 42nd ed. London, UK: Elsevier; 2021.
5. Haines DE, Mihailoff GA. Fundamental Neuroscience for Basic and Clinical Applications. 5th ed. Philadelphia, PA: Elsevier; 2018.
6. Guyton AC, Hall JE. Textbook of Medical Physiology. 14th ed. Philadelphia, PA: Elsevier; 2021.
7. Purves D, Augustine GJ, Fitzpatrick D, et al. Neuroscience. 6th ed. New York, NY: Oxford University Press; 2018.
8. Felten DL, Maida MS. Netter’s Atlas of Neuroscience. 4th ed. Philadelphia, PA: Elsevier; 2021.
9. Wolf, RK; Miranda, EA. Minimally Invasive Surgical Treatment of Atrial Fibrillation: A New Look at an Old Problem. 2024 Operative Techniques in Thoracic and Cardiovascular Surgery.
10. Takotsubo Cardiomyopathy Harvard.edu
- Details
- Written by: Efrain A. Miranda, Ph.D.
- Hits: 5220
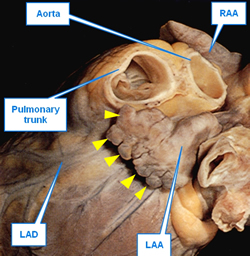
UPDATED: The left atrial appendage (LAA) is an embryological remnant of the development of the heart. It represents the primitive left atrium (LA) which is then “pushed to the side” by the development of the final (adult) stage of the LA. While the LAA is thin, tubular, tortuous, and presents with convoluted muscular walls, the adult LA has smooth walls and is considered to be a dilation of the terminal portion of the veins that enter the LA, hence the name “sinus venarum”, another term for the atria. The LAA is also known as the "left atrial appendix", or the "left auricle".
The anatomy of the LAA is presented in the video included at the end of the article, but there are some details that are important to discuss in the involvement of the LAA in the creation of thrombi and emboli in the presence of atrial fibrillation (AFib).
LAA shape and size
The LAA has important anatomical variations, with different shapes that anatomists and physicians have tried to consolidate in groups such as: chicken wing, cactus, windsock, cauliflower, etc. The fact is that recording the shape of the LAA is subjective. as the evaluation depends completely on the observer.
Researchers have tried to determine what shape can lead to a higher potential for stroke-producing emboli when AFib is present. A recent study by Dudzińska-Szczerba (2021) and an editorial by Yong Shin (2021) states that the shape itself is not a good predictor, but the distance between the LAA ostium and the first bend of the LAA is indeed a good predictor. The longer the distance there is increased potential for thrombus and emboli formation.
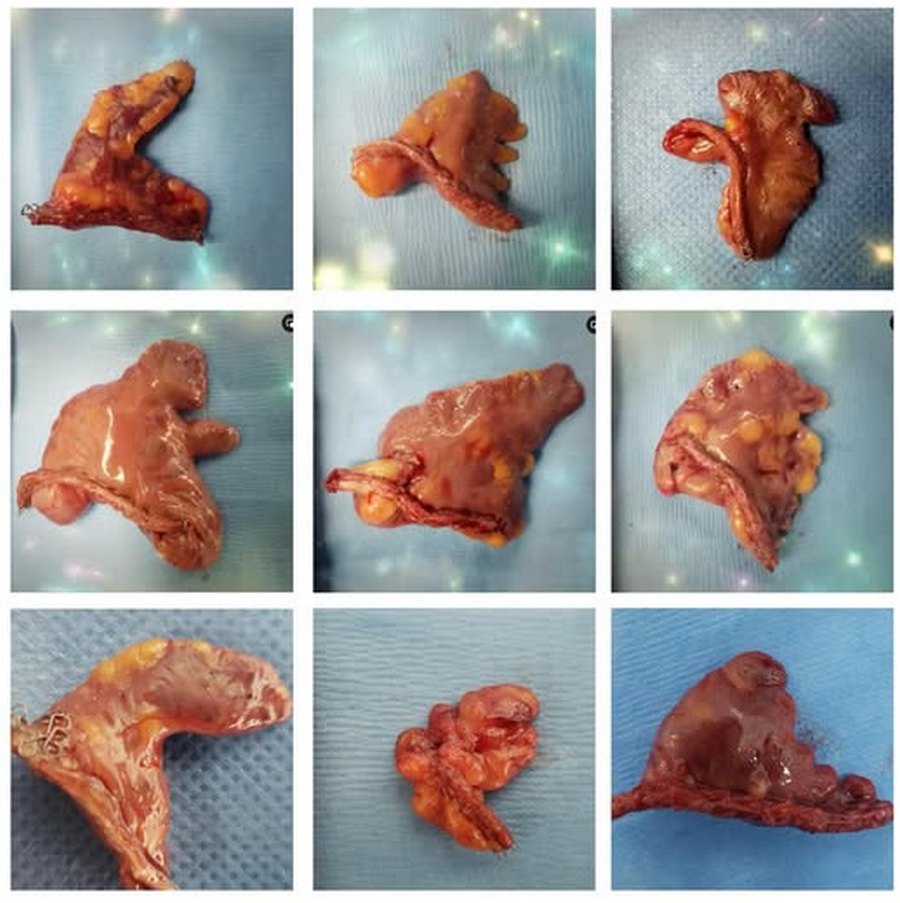
Left atrial appendage shapes. Courtesy of Sandra Fezzat Schrameyer
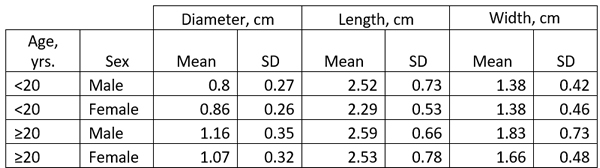
The size of the LAA has been studied in detail and it ranges ranging from 0.3 cm to 2.0 cm in males, and 0.3 to 1.8 cm in females. (Venoit, 1997), as shown in this table.
The LAA ostium
The LAA ostium is the communication between the LA and the LAA, it is generally oval in shape and its size is variable. The ostium is in some cases slit-like, or an elliptical-shaped variant, “smiley”, and even small circular (DeSimone, 2015; Cabrera, 2014). A study by Wang (2010) classified the LAA ostium into five types: oval (68.9%), foot-like (10%), triangular (7.7%), water drop-like (7.7%), and round (5.7%). It is interesting that devices that are used to occlude the LAA ostium are round and that is only 6% of the population reported in the Wang (2010) study. In a study by Su (2006) it was found that 100% of the specimens studied the LAA ostium had an oval shape with the mean diameter of the opening of 17.4 mm with a range between 10-24.1 mm).
In reference to LAA ostium occluders Su (2006) states that "These percutaneous devices / systems,however, have a round shape to fill or cover the LAA ostium. A previous study and our study show that the shape of the LAA ostium is consistently elliptical rather than round. This suggests that to seal the LAA orifice adequately without oversizing, devices may need to be elliptical for a snug fit. A round implant over an oval-shaped orifice may leave crevices on either side of the implant, leading to incomplete sealing of the orifice."
Lobes
The LAA can also present with different dilations called “lobes” these can range from zero to three or four.
Muscular Wall Structure
The LAA has internal ridges that form a muscular meshwork. The term used to describe these is “trabeculated”. It makes sense that in the case of atrial fibrillation, the slow to non-existent flow of blood within the deep recesses of the trabeculated muscular wall of the LAA will cause blood to pool and coagulate, forming thrombi. The presence of these LAA trabeculations have been found to be associated with stroke risk by Dudzińska-Szczerba (2021). The accompanying image shows the trabeculations in a cow's LAA. They are not as deep or as convoluted as those found in a human heart.
Crenellations
This is a rarely used term. It is a pattern along the top of a fortified wall, as in a castle, forming multiple, regular, rectangular spaces. These crenellations are found in the edge of the LAA compounding the irregularity of the internal wall and increasing the chance for thrombus formation and stroke-inducing emboli. Crenellations are shown by yellow triangles in the first image in this article.
Function of the LAA
As stated, the LAA is an embryological remnant, but it does have a function in the adult. It generates a peptide involved in the control of salt in the circulatory system. This is the atrial natriuretic peptide (ANP), a hormone that is secreted by both the right and left atria and their appendages in response to circulatory volume and pressure changes. ANP helps the elimination of excess sodium through the kidneys (natriuresis), control of urine elimination (diuresis), and antifibrotic and antihypertrophic effects within the heart (Sandeur, 2023)
While removing both the right and left atrial appendages could cause ANP deficiency, surgical removal or exclusion of only the LAA does not cause an ANP problem (8).
Involvement of the LAA in AFib
The LAA is an electrically active structure. The cardiomyocytes that form its walls have automatic activity and it has been described as an area that can trigger AFib. The accompanying video shows an LAA that has been separated from the heart (in this case using a surgical stapler) and it can be seen how the LAA continues fibrillating on its own. Video courtesy of Dr. Randall K. Wolf
This is the why LAA exclusion is a must in the case of AFib and potentially in any cardiovascular procedure where the pericardial sac is opened (this is a subject for discussion).
The problem is that devices that only occlude the LAA ostium do not electrically isolate the LAA wall from the LA wall, leaving this potential AFib-producing connection intact. Just occluding the LAA ostium is not a solution for atrial fibrillation, it just reduces the risk of stroke.
Personal note: In May 5th, 2020 Dr. Randall K. Wolf invited me to a live webcast where we reviewed the anatomy of the left atrial appendage, the problems the LAA can cause in atrial fibrillation leading to stroke, and the reasons for its exclusion in AFib surgery. This video is next. You can watch other videos on the topic here. Dr. Miranda.
Sources:
1. “Anatomy of the Normal Left Atrial Appendage: A Quantitative Study of Age-Related Changes in 500 Autopsy Hearts: Implications for Echocardiographic Examination” Veinot, JP; et al. 1997 Circulation; 96:3112–3115
2. “A Review of the Relevant Embryology, Pathohistology, and Anatomy of the Left Atrial Appendage for the Invasive Cardiac Electrophysiologist” De Simone, CV, et al. J AFib 2015; 8:2 81-87
3. “Left atrial appendage: anatomy and imaging landmarks pertinent to percutaneous transcatheter occlusion” Cabrera,JA; Saremi, F; Sanchez-Quintana, D. 2014 Heart 2014 100:1636-1650
4. Left Atrial Appendage Studied by Computed Tomography to Help Planning for Appendage Closure Device Placement” Wang Y. et al. J Cardiovasc Electrophyisiol 2010 21:9 973-982
5. IIs the Left Atrial Appendage (LAA) anatomical shape really meaningless measure for stroke risk assessment? Shin, SS; Park, JW. Int J Cardiol 2021 May 1:330:80-81. doi: 10.1016/j.ijcard.2021.02.047.6. “Assessment of the left atrial appendage morphology in patients after ischemic stroke” Dudzińska-Szczerba, K. et al. Int J Cardiol 2021 330:65-72
7. “Atrial Natriuretic Peptide” Sandeur, CC; Jialal, I. Stat Pearls 2023. StatPearls https://www.ncbi.nlm.nih.gov/books/NBK562257/
8. Personal communication, Dr. R. Wolf 2023
9. "Slide Atlas of Human Anatomy" Gosling, J.A.; Whitmore, I; Harris, P.F.; Humpherson, J.R., Et al; ISBN: 0723426570 Hong Kong: Times Mirror, 1996
10. "Atrial and brain natriuretic peptides: Hormones secreted from the heart" Nakagawa Y, Nishikimi T, Kuwahara K. Peptides. 2019 Jan;111:18-25.
11. "Occluding the left atrial appendage: anatomical considerations" Su, P; McCarthy, KP; Ho, SY. 2008 Heart 94:1166–1170
Thanks to Sandra Fezzat Schrameyer for the image of the different shapes of the LAA.